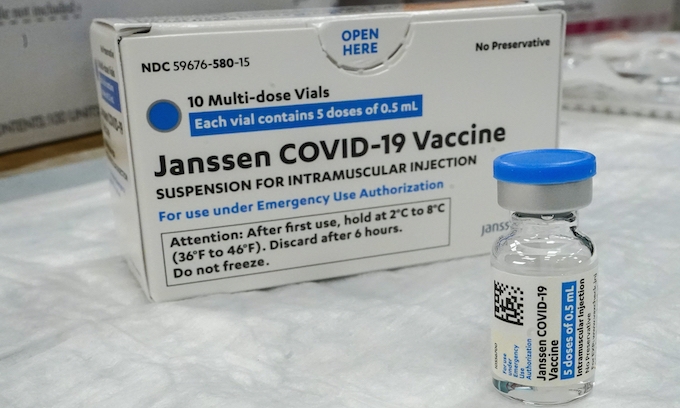
The approved shelf life of Johnson & Johnson’s one-and-done COVID-19 vaccine has been extended by 50% amid reports millions of unused doses were nearing expiration, the company confirmed Thursday.
The Food and Drug Administration granted the new authorization after “data from ongoing stability assessment studies” proved that doses were still stable and effective 4½ months after packaging when properly refrigerated, the company said.
The single-shot vaccine previously had an approved shelf life of only three months.
The 45-day extension comes at a critical time for the U.S. vaccination effort, with health officials scrambling to reach President Biden’s goal of at least partially vaccinating 70% of Americans by July 4.
A combination of softening demand for COVID vaccines overall and lingering safety concerns about the J&J shot, which was linked to extremely rare blood clots, left nearly half of the 21 million doses the company produced for the U.S. sitting unused, Reuters reported.
At least 13 lots of J&J shots carried expiration dates of June 27 or earlier, Reuters said.
The J&J shot is considered an important option in the global fight against COVID-19 because it’s easier to distribute to underserved and remote locations than competing mRNA vaccines, which require more intense refrigeration and two doses spaced weeks apart.
©2021 New York Daily News. Visit nydailynews.com. Distributed by Tribune Content Agency, LLC.
—-
This content is published through a licensing agreement with Acquire Media using its NewsEdge technology.












You can pick it up in your grocers dairy section between the sour cream and cottage cheese.
Except that they don’t dare change the expiration dates on the sour cream and cream cheese.
I’ve actually shopped in a store, where one of the NICE things they used to do, was if you found Expired food, you could bring it up to the front desk, and they’d actually GIVE YOU a voucher for the price of that item.. So, if say you found 20 bucks worth of expired canned goods, 8 bucks in cheese stuff, and 12 bucks of meat, you’d get a voucher for 40 bucks…
HOWEVER iirc, they stopped that practice, after they had over a Dozen of their stores literally raided into by entire families of immigrants/welfare leeches, who’d spend 2+ hours, in the store going over EVERYTHING and raking up hundreds of bucks in free stuff….
These vaccines are causing heart problems in young people, but Joe is pushing all of the vaccines for young people, who have little risk for the disease.
Gotta keep Big Pharma happy, or NO political donations .
HENCE WHY I say get RID OF allowing any company to donate, UNLESS ITS DONE BY INDIVIDUALS THEMSELVES..